Fuel Cell Technologies - An External Supply of Chemical Energy
06 Mar 2018
Generating Electricity from an Electrochemical Reaction
A fuel cell is like a battery in that it generates electricity from an electrochemical reaction. Both batteries and fuel cells convert chemical potential energy into electrical energy and also, as a by-product of this process, into heat energy. However, a battery holds a closed store of energy within it and once this is depleted the battery must be discarded or recharged by using an external supply of electricity to drive the electrochemical reaction in the reverse direction.
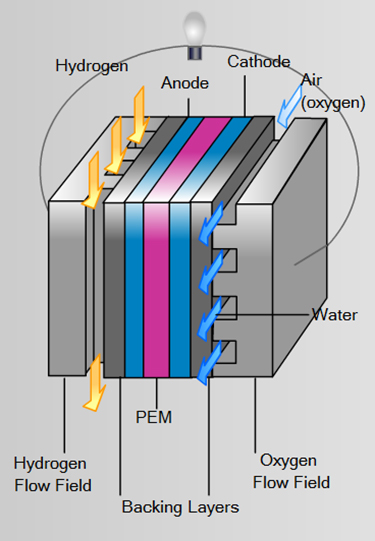
Electrochemical Reactionsin a Fuel Cell
Cathode: O2 + 4H+ + 4e– → 2H2O
Anode: 2H2 → 4H+ + 4e–
Overall: 2H2 + O2 → 2H2O
A fuel cell, on the other hand, uses an external supply of chemical energy and can run indefinitely, as long as it is supplied with a source of hydrogen and a source of oxygen (usually air). The source of hydrogen is generally referred to as the fuel and this gives the fuel cell its name, although there is no combustion involved. Oxidation of the hydrogen instead takes place electrochemically in a very efficient way. During oxidation, hydrogen atoms react with oxygen atoms to form water; in the process electrons are released and flow through an external circuit as an electric current.
Fuel cells can vary from tiny devices producing only a few watts of electricity, right up to large power plants producing megawatts. All fuel cells are based around a central design using two electrodes separated by a solid or liquid electrolyte that carries electrically charged particles between them. A catalyst is often used to speed up the reactions at the electrodes. Fuel cell types are generally classified according to the nature of the electrolyte they use. Each type requires particular materials and fuels and is suitable for different applications.
There are six major types of fuel cells, namely Proton Exchange Membrane Fuel Cell (PEMFC), Direct Methanol Fuel Cells (DMFC), Solid Oxide Fuel Cells (SOFC), Alkaline Fuel Cells (AFC), Molten Carbonate Fuel Cells (MCFC) and Phosphoric Acid Fuel Cells (PAFC).
Fuel cells are used today in a range of applications, from providing power to homes and businesses, to recharging consumer electronics, to moving a variety of vehicles including warehouse forklift fleets, buses, trucks, cars, and more. Today fuel cells are being used in many applications such as stationary power, vehicles and materials handling.
Current challenges in fuel cells include high cost of precious metal based electrocatalysts, carbon corrosion in PEM fuel cells, impurities in the fuel and air – starting and stopping (cycles), freezing and thawing, and humidity plus load cycles that result in stresses on the chemical and mechanical stability of the fuel cell system materials and components.
Intertek offers the following consulting services for customers who manufacture or utilize fuel cells:
- Reliability, safety and failure analysis
- Failure Mode and Effects Analysis (FMEA)
- Specification Development
- Technical Due Diligence
- Modeling and Simulation of Components and Systems
- Warranty Investigations
- Training and Courses
- On-Site Expert Resources
To learn more, visit our website or contact us.
The goal of Asset Integrity Management (AIM) is to effectively manage corporate assets in order to gain maximum value, profitability and returns while safeguarding personnel, the community, and the environment. At Intertek our AIM team delivers trusted and innovative technical solutions that ensure the quality, safety and reliability of our clients' assets.
