Our shared audits or joint audits for pharmaceutical or cosmetic supply chains provide a cost-effective solution for the audit of suppliers or subcontractors
Pharmaceutical shared audits bring cost efficiencies, streamline audit scheduling, and can help to facilitate greater collaboration with your suppliers. Our shared audit team are a global network of highly trained professionals who have both local knowledge and an in-depth understanding of the relevant national and international regulations.
What is a shared audit?
GXP Pharmaceutical supply chain auditing is a key requirement in the pharma sector where is an obligation to engage with supply chain members on matters concerning quality. A shared audit of suppliers (or joint audit) means the audit is sponsored by more than one pharmaceutical manufacturer for their collective use by combining the requests to audit a particular supplier site.
What are the benefits of a shared audit?
Shared audit programmes allow multiple drug manufacturers to evaluate the same supplier simultaneously through a single third-party audit while maintaining the same focus on quality and attention to detail. This results in both lower audit costs for pharmaceutical companies and fewer audits for each supplier to host, reducing demands on time and resources for all stakeholders and boosting cost efficiencies whilst enabling smoother audit process and scheduling of audit dates. Shared Audits also provide insight into the supply chain, trends and areas improvements are required. As shared supplier quality audits are conducted by a single third-party audit professional, a consistent supplier audit process approach provides a clearer understanding of what is expected of the supplier whilst still providing a relevant audit report.
Why choose a third-party shared audit?
Managed shared audits are growing in popularity as suppliers and subcontractors are increasingly being asked to take part in multiple audits which each demand precious time and resources. It is, however, wise for a pharmaceutical manufacturer to consider using a mix of individual and joint audits as appropriate to their needs. Shared audits are ideal for conducting a routine periodical assessment of an existing supplier (involving a general audit of a supplier’s quality systems) or can be suitable where the service or product could be considered off-the-shelf or can also be used as part of the screening process for potential suppliers.
Our Shared Audits Drive Pharmaceutical Supply Chain Visibility
Intertek’s pharmaceutical shared audit team simplify the process of audit scheduling and help to facilitate greater collaboration with your suppliers and expedites supply chain visibility. You will receive the exact information to meet your auditing requirements via a personalised audit report whilst benefitting from the cost efficiencies of the shared audit process. Added benefits include evaluation of the supplier’s action plan from the auditor, on-site monitoring of the action plan implementation and technical support during the audit.
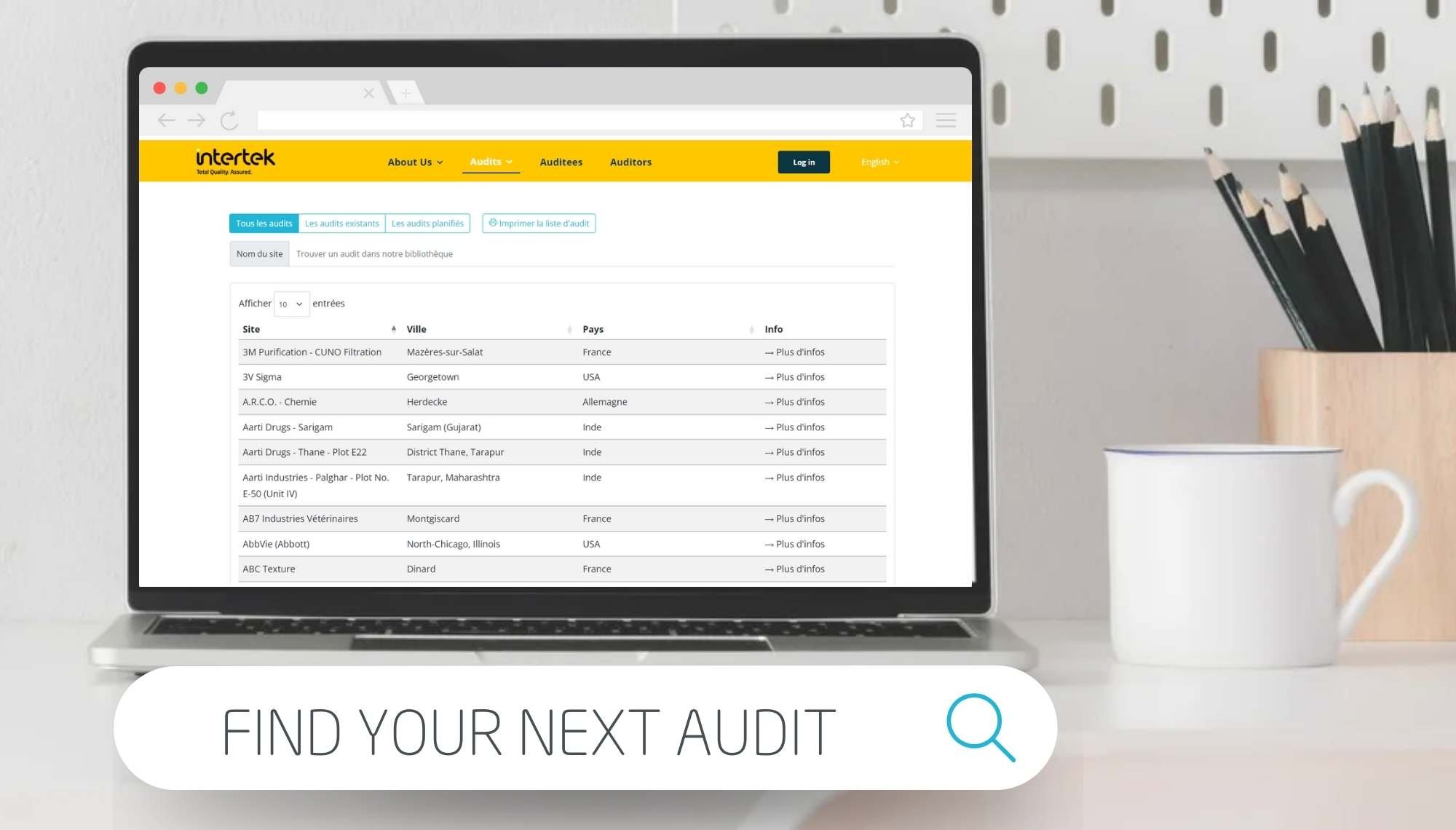
Intertek's Shared Audits Platform
Our confidential shared Audit Live List and shared audit system allow access to a real-time view of Intertek’s forthcoming scheduled GXP shared audits of manufacturers and distributors which will be conducted by our independent, qualified and highly experienced auditors including:
- Suppliers, APIs, excipients, cosmetic ingredients, starting materials, packaging materials and more
- Contract Manufacturing Organizations, Pharmaceutical, Cosmetic, Combination Products and Nutritional Supplements.
- Contract Research Organizations: Clinical Research Organisations, Contract fill and finish and more.
After logging in to your secure account, you can view details of past audits for specific plants, view requests for audits and apply to participate in a shared audit. The shared audit system allows a simple means to initiate and manage your required supplier audits. Our shared audits are conducted following relevant international standards and are specific to the type of audit activity required. Our qualified auditors conduct audits with reference to cGMP, GDP, GLP & GCP, ISO 22716, ISO 13485, IPEC, EFfCI standards within a European, North American, Japanese, or Australian context. We have 15 years of international experience in auditing and evaluating production and distribution sites and so contact our expert audit team now to find out how our shared audit services can benefit your business.
Audit Report Purchase Requests
In some cases, we can offer the purchase of an existing GMP, GDP or GCP audit report. This is a confidential process which enables us to explore, on your behalf, the potential purchase of an existing audit report. This represents cost savings to both the audit report owner and enquirer.
Total Quality Assurance
We are ISO 9001 certified. Quality is at the heart of our organisation and we continuously focus on improving the performance of our services in order exceed expectations of our global clients. With over 1600 global clients who put their trust in us to meet their auditing requirements, our shared audit system and services provide cost-effective and responsive auditing solutions for the global pharmaceutical and healthcare industries. Our Total Quality Assurance expertise offers you a truly tailored and unique service that will meet your specific needs.
Subscribe now
Subscribe to the Intertek Shared Audit newsletter