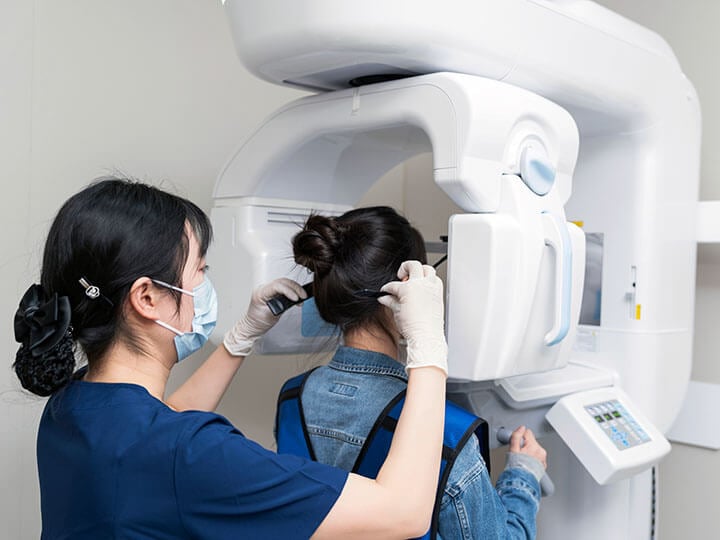
IEC 60601 is a widely accepted series of international standards for the Basic Safety and Essential Performance of Medical Electrical Equipment
What is IEC 60601?
IEC 60601 is a series of international standards, published by the International Electrotechnical Commission (IEC), that specify safety and performance requirements for medical electrical equipment and is widely recognized as the benchmark for medical device safety. The IEC 60601 series consists of multiple parts, each focusing on different aspects of medical electrical equipment.
Benefits of IEC 60601
One of the primary goals of IEC 60601 is to ensure the safety of patients using medical electrical equipment. Compliance with IEC 60601 standards helps to minimize the risk of electrical hazards, such as electric shocks or burns, ensuring that devices are designed and manufactured with patient safety as a top priority. The IEC 60601 standards also provide a consistent framework for safety requirements across different types of medical electrical equipment. This uniformity helps manufacturers understand and implement the necessary safety measures, leading to a higher level of safety across the industry.
Many countries have adopted the IEC 60601 series as the basis for their medical device regulations. Compliance with these standards is often a legal and regulatory requirement for manufacturers to demonstrate the safety and performance of their devices. Adhering to IEC 60601 helps manufacturers meet these regulatory obligations and gain market access in various countries.
Intertek Solutions for IEC 60601
As a Nationally Recognized Testing Laboratory (NRTL) approved by OHSA, Intertek is an industry leader providing Testing, Certification and in-lab support to help you navigate the requirements of the IEC 60601 series of standards. With our team of experts globally, Intertek is positioned to help determine standard applicability and execution. Through standards testing, you can support your safety claims throughout North America, Europe and beyond. We offer end-to-end solutions from product development, to review of the Risk Management File and supporting documentation, to comprehensive testing to the 60601/80601 series of standards, including an array of Collateral (IEC 60601-1-X) and Particular (IEC 60601-2-X) Standards. Intertek is your trusted partner throughout the testing process.
- IEC 60601-1-2: Medical Device EMC Testing
- IEC 60601-1-9: Requirements for Environmentally Conscious Design
- ISO 14971: Application of Risk Management for Medical Devices
- Customized On-site & Off-site Training Seminars
- Testing and Certification to IEC/AAMI 60601-1, 3rd Edition including Amendment 1 and 2
- Differences between the EU MDD and the MDR - FAQ Download
IEC 60601 Resources
Knowledge Center
Evolution of the Home Healthcare Market and Medical Device Compliance
White Paper | Webinar Recording
Ventilator Production: Introductory Guide to Regulatory Requirements
Critical Care Medical Devices: First-In-Queue Priority
Product Listing & Marking Strategies White Paper
IEC 60601-1-2 Edition 4 White Paper
Updated FCC Approval Process Webinar
Extractables and Leachables Studies Fact Sheet
Five Steps to Medical Device Commercialization in the United States White Paper
Making Green Profitable: Using IEC 60601-1-9 as a Competitive Advantage White Paper
*The Intertek legal entities that provide medical device management system certification services (including ISO 13485 and MDSAP) and Notified Body services (MDR 2017/745 and MDD 93/42/EEC) do not provide any consulting services. Clients who have used other Intertek legal entities’ consulting services are not eligible to receive management system certification services or Notified Body services from Intertek.