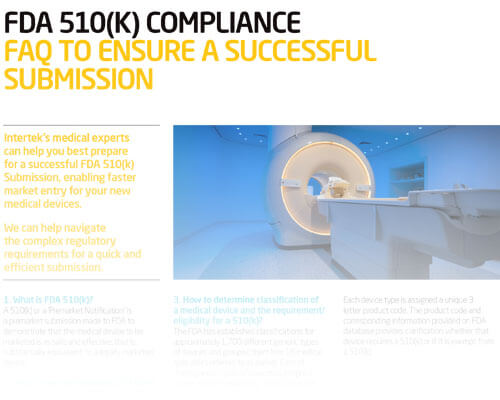
Preliminary Guidance for Building a Successful FDA 510(k) Submission
FDA clearance is critical to your market success and Intertek will help you navigate the 510(k) submission process quickly and efficiently.
Missing information and deficiencies in your 510(k) submission can lead to failure and may delay your product launch by months. Intertek Consultants provide one-on-one preliminary guidance for your 510(k) submission, helping you shorten your certification process and get to market on time. Our lead consultant has worked directly with FDA branch chiefs and reviewers, which translates into extensive product knowledge and industry experience passed on to you throughout your 510(k) submission review.
Initiate your 510(k) Consulting Project
Download Form Now
Knowledge Center
Evolution of the Home Healthcare Market and Medical Device Compliance
White Paper | Webinar Recording
Ventilator Production: Introductory Guide to Regulatory Requirements
Critical Care Medical Devices: First-In-Queue Priority
Product Listing & Marking Strategies White Paper
IEC 60601-1-2 Edition 4 White Paper
Updated FCC Approval Process Webinar
Extractables and Leachables Studies Fact Sheet
Five Steps to Medical Device Commercialization in the United States White Paper
Making Green Profitable: Using IEC 60601-1-9 as a Competitive Advantage White Paper
Related Links
- ETL Mark The Industry's Fastest Certification Program
- Search and Buy Medical Device Standards
- Reese's Law – ANSI/UL 4200A-2023
- My Test Central
- Directories
- Certification Marks
- Global Research & Certification
- Medical Podcast - Compliance with Clarissa
- Satellite Data Acceptance Program
- Planning for Quality through Disruptions
- Intertek's world-class team of Medical Experts
- Intertek Protek - The world’s first industry-agnostic, end-to-end health, safety and wellbeing assurance program
Intertek does not provide consulting services for management systems certification. Any consulting activities provided by Intertek are separate and independent from certification activities.